Research Description
Research Interest
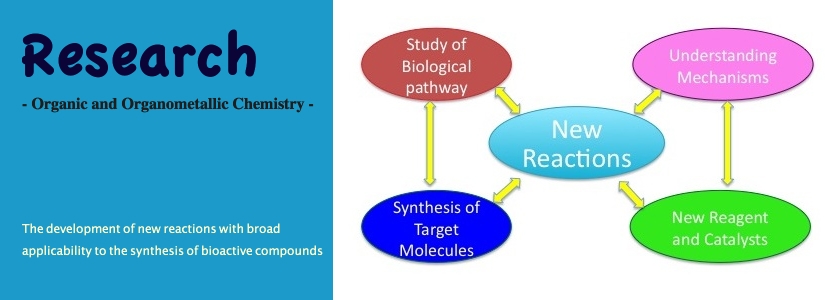
Our primary research interests lie in the areas of organic synthesis and reaction methodology. The development of new carbon-carbon bond-forming reactions with broad applicability to the synthesis of biologically significant targets will be the primary focus of our research group.
Methodology Development
The center of our research activity is new reaction development. Our research projects will encompass new reaction development including new catalysts, elucidation of reaction mechanisms and application of the new methodologies to the synthesis of bioactive compounds. Organic synthesis shows an enormous potential to produce chemotherapeutic agents, and chemical reactions are one of the most powerful tools for the drug discovery process.Currently we are focusing on quinones and indole quinones. Also, the compounds obtained in the course of methodology developments will be tested in bioactive assays in a future collaboration with biology faculty at UTRGV.
Synthesis of small natural products
Nature can generate complex structures with simple reactions. We are attempting to investigate some of these biochemical processes, and understand the mechanisms that they follow. We are presently working on the synthesis of small natural products which demonstrate anticancer activities.
Synthesis under the sun
The Sunlight drives a large number of chemical reactions for life on the earth, and it is a unique natural resource, which is renewable as a clean energy and has huge potential for valuable applications. Solar radiation have attracted rising attention in scientific society for sustainable and environmentally friendly technologies. In synthetic organic photochemistry, artificial UV light has been used predominantly, but reactions driven by visible light have started to be explored in recent years. In our group, quinone derivatives have been selected as the substrates due to their great abilities as electron acceptors. Also, quinones are widely found in nature and drugs such as anthracyclines, daunorubicin, mitomycin, which are clinically used in the therapy of solid cancers. Thus, we are trying to work on the new synthesis of quinone derivatives under the sun.
Those who love organic chemisrty are very welcome to join us! Contact Dr. Mito for more information!

 Home
Home